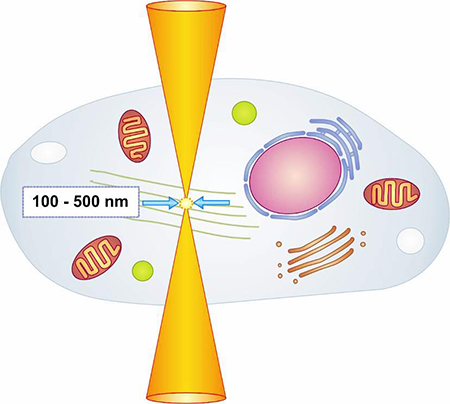
1. Nolte, P.; Brettmacher M.; Gröger, C. J.; Gellhaus, T.; Svetlove A.; Schilling, A. F.; Alves, A.; Rußmann, C.; Dullin, C.; (2023) Spatial correlation of 2D hard‑tissue histology with 3D microCT scans through 3D printed phantoms Sci Rep 13, 18479
2. Kevin Janot, Grégoire Boulouis, Géraud Forestier, Fouzi Bala, Jonathan Cortese, Zoltán Szatmáry, Sylvia M. Bardet, Maxime Baudouin, Marie-Laure Perrin, Jérémy Mounier, Claude Couquet, Catherine Yardin, Guillaume Segonds, Nicolas Dubois, Alexandra Martinez, Pierre-Louis Lesage, Yong-Hong Ding, Ramanathan Kadirvel , Daying Dai, Charbel Mounayer, Faraj Terro, Aymeric Rouchaud. (2023) WEB shape modifications: “angiography–histopathology correlations in rabbits” J NeuroIntervent Surg 2023;0:1–7.
3. Géraud FORESTIER, Jonathan CORTESE, Sylvia M. BARDET, Maxime BAUDOUIN, Kévin JANOT, Voahirana RATSIMBAZAFY, Marie-Laure PERRIN, Jérémy MOUNIER, Claude COUQUET, Catherine YARDIN, Yan LARRAGNEGUY, Flavie SOUHAUT, Romain CHAUVET, Alexis BELGACEM, Sonia BRISCHOUX, Julien MAGNE, Charbel MOUNAYER, Faraj TERRO, Aymeric ROUCHAUD. (2023) “Comparison of Arterial Wall Integration of different Flow Diverters in rabbits” the CICAFLOW study Journal of Neuroradiology, In press.
4. Donath, Sören, Leon Angerstein, Lara Gentemann, Dominik Müller, Anna E. Seidler, Christian Jesinghaus, André Bleich, Alexander Heisterkamp, Manuela Buettner, and Stefan Kalies. (2022). “Investigation of Colonic Regeneration via Precise Damage Application Using Femtosecond Laser-Based Nanosurgery” Cells 11, no. 7: 1143. https://doi.org/10.3390/cells11071143
5. Müller, Dominik, Sören Donath, Emanuel G. Brückner, Santoshi Biswanath Devadas, Fiene Daniel, Lara Gentemann, Robert Zweigerdt, Alexander Heisterkamp, and Stefan M.K. Kalies. (2021). “How Localized Z-Disc Damage Affects Force Generation and Gene Expression in Cardiomyocytes” Bioengineering 8, no. 12: 213. https://doi.org/10.3390/bioengineering8120213
6. Müller D, Klamt T, Gentemann L, Heisterkamp A, Kalies SMK (2021) Evaluation of laser induced sarcomere micro-damage: Role of damage extent and location in cardiomyocytes. PLoS ONE 16(6): e0252346. https://doi.org/10.1371/journal.pone.0252346
7. Bouyer M; Garot C; Machillot P; Vollaire J; Fitzpatrick V; Morand S; Boutonnat J; Josserand V; Bettega G; Picart C (2021) 3D-printed scaffold combined to 2D osteoinductive coatings to repair a critical-size mandibular bone defect Materials Today Bio 11 100113
8. Verhaegen C, Kautbally S, Zapareto D C, Brusa D, Courtoy G, Aydin S, Bouzin C, Oury C, Bertrand L, Jacques P J, Beauloye C, Horman S, Kefer J (2020) Early thrombogenicity of coronary stents: comparison of bioresorbable polymer sirolimus-eluting and bare metal stents in an aortic rat model. Am J Cardiovasc Dis. 10(2):72-83
9. Zeller-Plumhoff B, Malicha C, Krüger D, Campbella G, Wiesea B, Galli S, Wennerberg A, Willumeit-Römer R, Wieland F (2020) Analysis of the bone ultrastructure around biodegradable Mg–x Gd implants using small angle X-ray scattering and X-ray diffraction Acta Biomaterialia 101 637–645
10. Rousselle S D , Wicks J R, Tabb B C, Tellez A, O’Brien M (2019) Histology Strategies for Medical Implants and Interventional Device Studies Toxicologic Pathology Vol. 47(3) 235-249
11. Neuerburg C, Mittlmeier L M, Keppler A M, Westphal I, Glass Ä, Saller M M, Herlyn P K E, Richter H, Böcker W, Schieker M, Aszodi A, Fischer D C (2019) Growth factor-mediated augmentation of long bones: evaluation of a BMP-7 loaded thermoresponsive hydrogel in a murine femoral intramedullary injection model. Journal of Orthopaedic Surgery and Research 14 297
12. Kunert-Keil C, Richter H, Zeidler-Rentzsch I, Bleeker I, Gredes T (2019) Histological comparison between laser microtome sections and ground specimens of implant-containing tissues. Annals of Anatomy 222 153–157
13. Gabler C, Saß JO, Gierschner S, Lindner T, Bader R, Tischer T (2018) In Vivo Evaluation of Different Collagen Scaffolds in an Achilles Tendon Defect Model. BioMed Research International 208
14. Wolkers W, Vásquez-Rivera A, Oldenhof H, Dipresa D, Goecke T, Kouvaka A, Will F, Haverich A, Korossis S, Hilfiker A (2018) Use of sucrose to diminish pore formation in freeze-dried heart valves. Scientific Reports 8 12982
15. Albers J, Markus MA, Alves F, Dullin C (2018) X-ray based virtual histology allows guided sectioning of heavy ion stained murine lungs for histological analysis. Scientific Reports 8(1) 7712
16. Boyde A (2018) Evaluation of laser ablation microtomy for correlative microscopy of hard tissues. Journal of Microscopy 271(8) 1-14
17. Pobloth AM, Checa S, Razi H, Petersen A, Weaver JC, Schmidt-Bleek K, Windolf M, Tatai AÁ, Roth CP, Schaser KD, Duda GN, Schwabe P (2018) Mechanobiologically optimized 3D titanium-mesh scaffolds enhance bone regeneration in critical segmental defects in sheep. Science Translational Medicine 10 423
18. Joner M, Nicol P, Rai H, Richter H, Foin N, Ng J, Cuesta J, Rivero F, Serrano R, Alfonso F (2018) Very Late Scaffold Thrombosis: Insights from Optical Coherence Tomography and Histopathology. EuroIntervention 13(18)
19. Boyde A, Staines KA, Javaheri B, Millan JL, Pitsillides AA, Farquharson C (2017) A distinctive patchy osteomalacia characterises Phospho1 deficient mice. Journal of Anatomy 231 298-308
20. Kowtharapu BS, Marfurt C, Hovakimyan M, Will F, Richter H, Wree A, Stachs O, Guthoff RF (2017) Femtosecond laser cutting of human corneas for the subbasal nerve plexus evaluation. Journal of Microscopy 265(1) 21–26
21. Will F, Richter H (2015) Laser-based Preparation of Biological Tissue. Laser Technik Journal 12(5) 44-47
22. Richter H, Ratliff J, Will F, Stolze B (2015) Time- and material saving laser microtomy for hard tissue and implants. European Cells and Materials 29 Suppl.2 4
23. Richter H, Ramirez Ojeda DF, Will F (2014) Lasergesteuerte Probenpräparation von Hartgeweben und Biomaterialien. BIOspektrum 05 14
24. Bourassa D, Gleber S-C, Vogt S, Yi H, Will F, Richter H, Shin CH, Fahrni CJ (2014) 3D Imaging of Transition Metals in the Zebrafish Embryo by X-ray Fluorescence Microtomography. Metallomics 6 1648-1655
25. Schimek K, Busek M, Brincker S, Groth B, Hoffmann S, Lauster R, Lindner G, Lorenz A, Menzel U, Sonntag F, Walles H, Marx U, Horland R. (2013) Integrating biological vasculature into a multi-organ-chip microsystem. Lab Chip 13 3588-3598
26. Richter H, Ratliff J (2012) A Non-Contact Method of Sectioning Cardiovascular Arteries Containing Metallic Stents Using Laser Technology. J Histotechnol 35 (4) 205
27. Richter H, Lubatschowski H, Will F (2011) Laser in Medizin & Biologie: Laser-Mikrotomie mit ultrakurzen Pulsen – Neue Perspektiven für die Gewebe- und Biomaterialbearbeitung. Biophotonik 09 50-52
28. Lubatschowski H, Will F, Przemeck S, Richter H (2011) Laser Microtomy. Handbook of Biophotonics Vol. 2: Photonics for Health Care Wiley-VCH 151-157
29. Kermani O, Will F, Massow O, Oberheide U, Lubatschowski H (2010) Control of Femtosecond Thin-flap LASIK Using OCT in Human Donor Eyes. Journal of Refractive Surgery 26(1) 57-61
30. Baumgart J, Bintig W, Ngezahayo A, Lubatschowski H, Heisterkamp A (2010) Fs-laser-induced Ca2+ concentration change during membrane perforation for cell transfection. Optics Express 18 (3) 2219
31. Kermani O, Will F, Massow O, Oberheide U, Lubatschowski H. (2009) Echtzeitsteuerung einer Femtosekundenlaser Sub-Bowman-Keratomileusis an humanen Spenderaugen mittels optischer Kohärenztomographie. Klin Monatsbl Augenheilkd 226 965-969
32. Kütemeyer K, Baumgart J, Lubatschowski L, Heisterkamp A (2009) Repetition rate dependency of low density plasma effects during femtosecond-laser-based surgery of biological tissue. Appl. Phys. B 97(3) 695
33. Baumgart J, Kuetemeyer K, Bintig W, Ngezahayo A, Ertmer W, Lubatschowski H, Heisterkamp A (2009) Repetition rate dependency of reactive oxygen species formation during femtosecond laser-based cell surgery. J Biomed Opt 14(5) 054040
34. Kermani O, Will F, Lubatschowski H (2008) Real-Time Optical Coherence Tomography-Guided Femtosecond Laser Sub-Bowman Keratomileusis on Human Donor Eyes. Am J Ophthalmol 146 42–45.
35. Kermani O (2008) „Sehendes Skalpell” schon heute realisierbar. Ophthalmologische Nachrichten 09 (Kongressausgabe)
36. Baumgart J, Bintig W, Ngezahayo A, Willenbrock S, Murua Escobar H, Ertmer W, Lubatschowski H, Heisterkamp A (2008) Quantified femtosecond laser based opto-perforation of living GFSHR-17 and MTH53a cells. Opt. Express 16(5) 3021-3031
37. Baumgart J, Kuetemeyer K, Bintig W, Ngezahayo A, Ertmer W, Lubatschowski H, Heisterkamp A (2008) Investigation of reactive oxygen species in living cells during femtosecond laser based cell surgery. Proc. SPIE Optical Interactions with Tissue and Cells XIX Vol 6854
38. Heisterkamp A, Baumgart J, Maxwell IZ, Ngezahayo A, Mazur E, Lubatschowski H (2007) Fs-Laser Scissors for Photobleaching, Ablation in Fixed Samples and Living Cells, and Studies of Cell Mechanics. Laser Manipulation of Cells and Tissues; Elsevier Inc. 293-307
39. Will F, Block T, Menne P, Lubatschowski H (2007) Laser Microtome: all optical preparation of thin tissue samples. Proceedings of SPIE 6460 646007-1
40. Lubatschowski H (2007) Laser Microtomy – Opening a new Feasibility for Tissue Preparation. Optic & Photonic WILEY-VCH 49 – 51
41. Menne P (2007) Microtomy with Femtosecond Lasers. Biophotonics International; Laurin Publishing Co. Inc. May 2007 35 – 37